A one pot synthesis of Au–ZnO nanocomposites for plasmon-enhanced sunlight driven photocatalytic activity†
Abstract
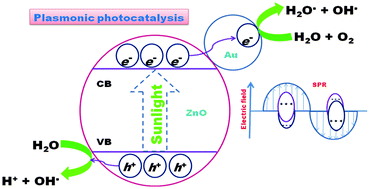
To enhance the photocatalytic efficiency of ZnO nanomaterials, plasmonic Au nanoparticles (NPs) have deliberately been introduced into ZnO through a facile, inexpensive one pot hydrothermal approach. The enhanced photocatalytic efficiency was ascribed to surface plasmon resonance induced local electric field enhancement of Au. Thus the photoproduced e−–h+ pair in ZnO under sunlight irradiation is reluctant to recombine. The as-synthesized photocatalyst with an excellent sunlight driven photocatalytic activity can effectively decompose various kinds of organic dyes and maintain a high level of photoactivity even after four cycles. The photocatalytic activity of the Au–ZnO nanocomposites was examined by the photodegradation of a series of cationic and anionic dye molecules such as rhodamine B, Congo red, methyl orange, methylene blue, and Rose Bengal. It is interesting to note that with a reasonable increase in the Au concentration the shape of the nanocomposites remains unaltered but the visible light driven photocatalytic activity is enhanced. This observation and the result are promising for plasmonic photocatalysis. The presence of Au nanoparticles makes the Au–ZnO nanocomposite a superior photocatalyst over the ZnO nanomaterial and the nanocomposite presents altogether a different scenario. In a nut-shell the present study reports not only a new insight into the gram level synthesis of a plasmonic photocatalyst from one pot but also its application in waste water treatment through the degradation of toxic dye molecules upon direct sunlight exposure.

 Please wait while we load your content...
Please wait while we load your content...