Mechanisms of cell uptake and toxicity of the anticancer drug cisplatin
Abstract
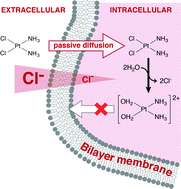
Two major issues which hamper the use of the anticancer drug cisplatin are the development of cancer cell resistance and its nephrotoxicity. One possible mechanism by which resistance is reported to develop is a reduction in drug uptake across the cell membrane. While the passive uptake of cisplatin has long been cited as an important contribution, far greater attention has been given to active modes of uptake, particularly in recent research. Using unilamellar lipid vesicles together with the stopped-flow kinetic method we show here that the permeability coefficient of cisplatin increases significantly with the chloride concentration of the medium. This supports the hypothesis that cisplatin can enter cells via passive permeation through the lipid phase of the membrane, but becomes trapped within the cytoplasm because dissociation of chloride ligands yields a membrane-impermeant positively-charged aqua derivative. This is important evidence for a major role of passive membrane diffusion in the uptake of cisplatin, and suggests that reduced cell uptake is unlikely to be a significant mechanism leading to the development of drug resistance. Studies of rubidium ion uptake into the cytoplasm of Xenopus oocytes via the Na+,K+-ATPase show significant inhibition of this ion pump when cisplatin is present in the cytoplasm. Because Na+,K+-ATPase activity is essential to the survival of all animal cells, e.g. via maintenance of cell volume, and the Na+,K+-ATPase is expressed at particularly high levels within the membranes of kidney tubules where it plays a crucial role in nutrient reabsorption, these results suggest that cisplatin-induced inhibition of the Na+,K+-ATPase is a likely contributing cause for the nephrotoxicity of cisplatin.

 Please wait while we load your content...
Please wait while we load your content...