On the catalytic mechanism of polysaccharide lyases: evidence of His and Tyr involvement in heparin lysis by heparinase I and the role of Ca2+†‡
Abstract
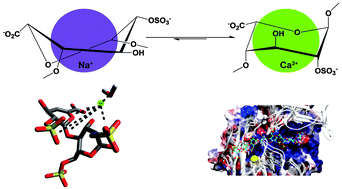
The structurally diverse polysaccharide lyase enzymes are distributed from plants to animals but share common catalytic mechanisms. One, heparinase I (F. heparinum), is employed in the production of the major anticoagulant drug, low molecular weight heparin, and is a mainstay of cell surface proteoglycan analysis. We demonstrate that heparinase I specificity and efficiency depend on the cationic form of the substrate. Ca2+–heparin, in which α-L-iduronate-2-O-sulfate residues adopt 1C4 conformation preferentially, is a substrate, while Na+–heparin is an inhibitor. His and Tyr residues are identified in the catalytic step and a model based on molecular dynamics and docking is proposed, in which deprotonated His203 initiates β-elimination by abstracting the C5 proton of the α-L-iduonate-2-O-sulfate residue in the substrate, and protonated Tyr357 provides the donor to the hexosamine leaving group.

 Please wait while we load your content...
Please wait while we load your content...