Surface modification of ionic liquid-spun chitin fibers for the extraction of uranium from seawater: seeking the strength of chitin and the chemical functionality of chitosan†
Abstract
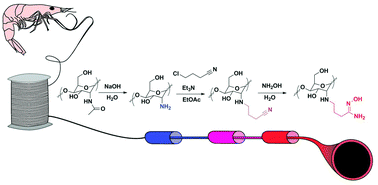
Chitin fibers, prepared by extracting chitin directly from shrimp shell waste and dry-jet wet spinning from the resulting ionic liquid (1-ethyl-3-methylimidazolium acetate) solution in a one-pot process, were surface modified by taking advantage of the insolubility of chitin in common solvents (e.g., water, organics). In this proof of concept example, the chitin fiber surfaces were first deacetylated using aqueous NaOH to make available the primary amine (the functional group of chitosan) on the surface. Further treatment of the fibers allowed for the task-specific tailoring of the functionality (here we appended amidoxime for the extraction of aqueous uranyl ions from seawater). Compositional analysis and physical property measurements (e.g., tensile strength and thermal decomposition) of the fibers before and after surface modification indicated minimal change to the bulk material; however, spectroscopy and sorption studies of uranyl ions from aqueous solution demonstrated surface modification. The lower cost, one-pot process used in this study resulted in weak and brittle fibers, suggesting that additional purification of the chitin before pulling fibers will greatly improve the strength and utility of the resulting material. Overall, a platform has been developed for the surface modification of chitin fibers that provides both the physical properties of chitin and the functional properties of chitosan, resulting in an advanced material from a biorenewable resource with reduced chemical input.

 Please wait while we load your content...
Please wait while we load your content...