Transformations of oxidized multiwalled carbon nanotubes exposed to UVC (254 nm) irradiation†
Abstract
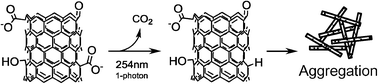
Motivated by the ability of UVC radiation to destroy harmful pathogens in drinking and waste water treatment plants, we have investigated the effect of 254 nm (UVC) radiation on the physical and chemical properties of oxidized multiwalled carbon nanotube (O-MWCNT) suspensions. Absorbance and particle size measurements were employed to monitor suspension stability, while X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), and Raman spectroscopy were used to characterize chemical and structural transformations. Experimental results indicate that after an initial period of irradiation where suspensions remained stable, exposure to 254 nm light caused O-MWCNT particles to aggregate. The duration of this initial period of particle stability increased at higher pH and lower ionic strength. This photo-induced aggregation was found to be the result of a loss of negatively charged carboxylic acid functional groups. Experiments performed in solutions containing different levels of dissolved oxygen, used to alter the concentration of reactive oxygen species (ROS) present during irradiation, indicate that ROS are not directly involved in the photodecarboxylation process. However, our results are consistent with a direct single photon decarboxylation process, previously identified in the organic photochemistry literature as the mechanism for CO2 removal from small organic molecules. Photodecarboxylation proceeded in the absence of any significant mass loss or changes in the O-MWCNT structure. Thus, UVC irradiation was unable to mineralize carbon atoms in the core of the O-MWCNTs, although it transformed the physical state of O-MWCNTs by changing the surface chemistry.

 Please wait while we load your content...
Please wait while we load your content...