Synthesis and characterization of isotopically labeled silver nanoparticles for tracing studies†
Abstract
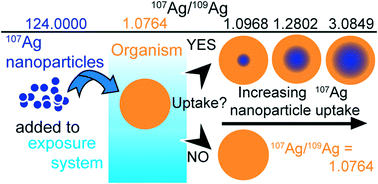
Silver nanoparticles (AgNPs) are ever more being used in industrial processes and consumer products, resulting in increasing emissions to the natural environment. To understand the behavior and environmental fate of AgNPs, it is paramount that they can be traced in complex natural samples from exposures at high sensitivity. The technique of stable isotope labeling is ideally suited for this purpose. To support such applications, we present a detailed evaluation of techniques for the preparation of stable isotope labeled AgNPs and demonstrate that isotopically modified particles are only distinguishable from particles with a natural isotope composition by their strong isotopic signature. Monodisperse suspensions of citrate-stabilized AgNPs with target sizes of 17, 20 and 30 nm were synthesized by reduction of silver nitrate solutions with sodium borohydride. The AgNP suspensions were produced using both natural Ag, which is comprised of the two stable isotopes 107Ag (52%) and 109Ag (48%), and Ag enriched to 99.2% in 107Ag. Synthesis was reliably reproduced on three separate occasions in two laboratories. The AgNPs were characterized using dynamic light scattering (DLS) shortly after synthesis and after up to 12 months storage. Some of the batches were also characterized using transmission electron microscopy (TEM) and asymmetric flow field-flow fractionation (FlFFF). The particle size distributions showed good reproducibility between the laboratories and stability over 12 months of storage. Importantly, the 107Ag-enriched particles were indistinguishable in size and shape from particles with a natural isotope composition. The reliability, control on particle size, and high yield of about 80%, demonstrate that the synthesis technique is well suited for small-scale production of isotopically labeled AgNPs. Isotope mass balance calculations furthermore show that the application of labeling enables tracing sensitivities for AgNPs that are at least 40 times, and possibly up to 4000 times, higher than those achievable with bulk Ag concentration measurements and experiments with exposure concentrations that approach predicted environmental levels are possible, if the most precise isotopic measurement techniques are employed.

 Please wait while we load your content...
Please wait while we load your content...