Stabilization of n-cadmium telluride photoanodes for water oxidation to O2(g) in aqueous alkaline electrolytes using amorphous TiO2 films formed by atomic-layer deposition†
Abstract
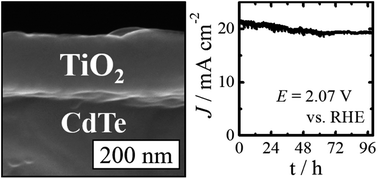
Although II–VI semiconductors such as CdS, CdTe, CdSe, ZnTe, and alloys thereof can have nearly ideal band gaps and band-edge positions for the production of solar fuels, II–VI photoanodes are well-known to be unstable towards photocorrosion or photopassivation when in contact with aqueous electrolytes. Atomic-layer deposition (ALD) of amorphous, “leaky” TiO2 films coated with thin films or islands of Ni oxide has been shown to robustly protect Si, GaAs, and other III–V materials from photocorrosion and therefore to facilitate the robust, solar-driven photoelectrochemical oxidation of H2O to O2(g). We demonstrate herein that ALD-deposited 140 nm thick amorphous TiO2 films also effectively protect single crystalline n-CdTe photoanodes from corrosion or passivation. An n-CdTe/TiO2 electrode with a thin overlayer of a Ni-oxide based oxygen-evolution electrocatalyst produced 435 ± 15 mV of photovoltage with a light-limited current density of 21 ± 1 mA cm−2 under 100 mW cm−2 of simulated Air Mass 1.5 illumination. The ALD-deposited TiO2 films are highly optically transparent and electrically conductive. We show that an n-CdTe/TiO2/Ni oxide electrode enables the stable solar-driven oxidation of H2O to O2(g) in strongly alkaline aqueous solutions, where passive, intrinsically safe, efficient systems for solar-driven water splitting can be operated.

 Please wait while we load your content...
Please wait while we load your content...