New insights into the electrochemical hydrogen oxidation and evolution reaction mechanism†
Abstract
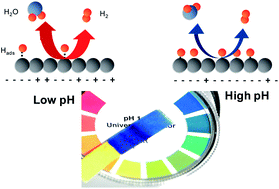
The effect of pH on the hydrogen oxidation and evolution reaction (HOR/HER) rates is addressed for the first time for the three most active monometallic surfaces: Pt, Ir, and Pd carbon-supported catalysts. Kinetic data were obtained for a proton exchange membrane fuel cell (PEMFC; pH ≈ 0) using the H2-pump mode and with a rotating disk electrode (RDE) in 0.1 M NaOH. Our findings point toward: (i) a similar ≈100-fold activity decrease on all these surfaces when going from low to high pH; (ii) a reaction rate controlled by the Volmer step on Pt/C; and (iii) the H-binding energy being the unique and sole descriptor for the HOR/HER in alkaline electrolytes. Based on a detailed discussion of our data, we propose a new mechanism for the HOR/HER on Pt-metals in alkaline electrolytes.

 Please wait while we load your content...
Please wait while we load your content...