Tri- and tetra-dentate imine vanadyl complexes: synthesis, structure and ethylene polymerization/ring opening polymerization capability†
Abstract
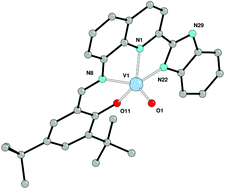
Reaction of the ligand 2,4-tert-butyl-6-[(2-methylquinolin-8-ylimino)methyl]phenol (L1H) with [VOCl3] in the presence of triethylamine afforded the complex [VOCl2L1] (1), whereas use of [VO(OnPr)3] led to the isolation of [VO2L1] (2) or [VO2L1]·2/3MeCN (2·2/3MeCN). Reaction of 2-((2-(1H-benzo[d]imidazol-2-yl)quinolin-8-ylimino)methyl)-4,6-R1,R2-phenols (R1 = R2 = tBu; L2H), (R1 = R2 = Me; L3H) or (R1 = Me, R2 = Ad; L4H) with [VO(OnPr)3] afforded complexes of the type [L2–4VO] (where L2 = 3, L3 = 4, L4 = 5). The molecular structures of 1 to 3 are reported; the metal centre adopts a distorted octahedral, trigonal bipyramidal or square-based pyramidal geometry respectively. In Schlenk line tests, all complexes have been screened as pre-catalysts for the polymerization of ethylene using diethylaluminium chloride (DEAC) as co-catalyst in the presence of ethyltrichloroacetate (ETA), and for the ring opening polymerization (ROP) of ε-caprolactone in the presence of benzyl alcohol. All pre-catalyst/DEAC/ETA systems are highly active ethylene polymerization catalysts affording linear polyethylene with activities in the range 3000–10 700 g (mol h bar)−1; the use of methylaluminoxane (MAO) or modified MAO as co-catalyst led to poor or no activity. In a parallel pressure reactor, 3–5 have been screened as pre-catalysts for ethylene polymerization in the presence of either DEAC or DMAC (dimethylaluminium chloride) and ETA at various temperatures and for the co-polymerization of ethylene with propylene. The use of DMAC proved more promising with 3 achieving an activity of 63 000 g (mol h bar)−1 at 50 °C and affording UHMWPE (Mw ∼ 2 000 000). In the case of the co-polymerization, the incorporation of propylene was 6.9–8.8 mol%, with 3 exhibiting the highest incorporation when using either DEAC or DMAC. In the case of the ring opening polymerization (ROP) of ε-caprolactone, systems employing complexes 1–5 were virtually inactive at temperatures <110 °C; on increasing the CL : V ratio at 110 °C, conversions of the order of 80% were achievable.

 Please wait while we load your content...
Please wait while we load your content...