In situ powder X-ray diffraction study of the hydro-thermal formation of LiMn2O4 nanocrystallites†
Abstract
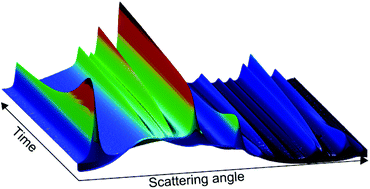
In situ measurements of the hydrothermal formation of LiMn2O4 (LMO) nanocrystallites reveal that the reaction progresses in steps, each creating a different crystalline phase. The reaction route is summarized as KMnO4→disordered δ-MnO2→(ordered δ-MnO2)→LiMn2O4→(γ-Mn2O3)→Mn3O4. The phase purity of LMO can be controlled by reaction time and temperature where phase pure LMO is obtained after 150–210 seconds at 220 °C or 45–140 seconds at 260 °C. It is also concluded that production of phase pure LMO by this method comes at the price of reduced reaction yield. From the observed reaction route an alternative way to control the phase purity is proposed by changing the amount of reducing agent. This hypothesis is rejected by a set of in situ measurements showing that the reaction kinetics of subsequent reaction steps hinders the formation of phase pure LMO. From the observation of unit cell changes as function of the transformation from LMO to Mn3O4 three distinct reaction parts are observed. This indicates that the reaction is a solid–solid reaction with a phase boundary. The in situ measurements reveal that LMO first appears in the reaction solution as thin platelets with sizes ranging from 3–13 nm. As the reaction progresses the crystallites grow faster along the [111] direction giving rod-like shaped crystallites in the end. The LMO crystallites start off with the same shape at all temperatures investigated indicating that they form from δ-MnO2 crystallites.
- This article is part of the themed collection: Inorganic Chemistry for Renewable Energy Conversion and Storage

 Please wait while we load your content...
Please wait while we load your content...