New supramolecular assemblies in heterobimetallic chemistry: synthesis of a homologous series of unsolvated alkali-metal zincates†
Abstract
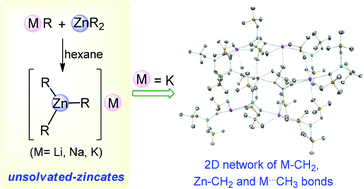
Using an interlocking co-complexation approach, a homologous series of unsolvated alkali-metal zincates [MZn(CH2SiMe3)3] (M = Li 1, Na 2, K 3) was prepared by reacting equimolar amounts of Zn(CH2SiMe3)2 with the relevant alkali-metal alkyl M(CH2SiMe3) employing non-coordinating hexane as a solvent. X-ray crystallographic studies reveal that these heterobimetallic compounds exhibit unprecedented supramolecular assemblies made up exclusively of a three-fold combination of M–CH2, Zn–CH2 and M⋯Me interactions. Revealing an important alkali-metal effect, 1 displays a linear chain structure; whereas 2 and 3 form much more intricate 3D and 2D coordination networks respectively. Shedding new light into the formation of these solvent-free zincates, DFT calculations indicate that the infinite degree of aggregation observed in 1–3 plays a major role in thermodynamically driving the co-complexation reactions of their homometallic precursors. NMR spectroscopic studies suggest that in C6D6 solution 1–3 exist as discrete contacted ion-pair species, where the alkali-metal is partially solvated by molecules of deuterated solvent. The supramolecular assemblies of 1–3 can be easily deaggregated by adding the polydentate N-donors PMDETA (N,N,N′,N′′,N′′-pentamethyldiethylenetriamine) or TMEDA (N,N,N′,N′-tetramethylethylenediamine), affording monomeric [(PMDETA)LiZn(CH2SiMe3)3] (4) and [(TMEDA)2NaZn(CH2SiMe3)3] (5).
- This article is part of the themed collection: New Expeditions in Polar Organometallic Chemistry

 Please wait while we load your content...
Please wait while we load your content...