Electronic tuning of Mo2(thioamidate)4 complexes through π-system substituents and cis/trans isomerism†
Abstract
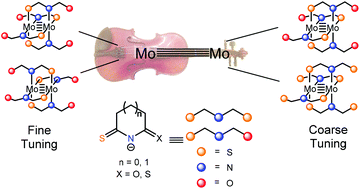
We report an exploration of the coordination chemistry of a systematic series of cyclic thioamidate ligands with the quadruply-bonded Mo24+ core. In addition to the S and N donor atoms that bind to Mo, the ligands utilized in this study have an additional O or S atom in conjugation with the thioamidate π system. The preparation of four new Mo2 complexes is described, and these compounds are characterized by X-ray crystallography, NMR and UV-vis spectroscopy, electrochemistry, and DFT calculations. These complexes provide a means to interrogate the electronics of Mo2(thioamidate)4 systems. Notably, we describe the first two examples of Mo2(thioamidate)4 complexes in their cis-2,2-regioisomer. By varying the π-system substituent and regioisomerism of these compounds, the electronics of the dimolybdenum core is shown to be altered with varying degrees of effect. Cyclic voltammetry results show that changing the π-system substituent from O to S results in an increase in the Mo24+/5+ oxidation potential by 170 mV. Changing the arrangement of ligands around the dimolybdenum core from trans-2,2 to cis-2,2 slightly weakens the metal–ligand bonds, raising the oxidation potential by a more modest 30–100 mV. MO diagrams of each compound derived from DFT calculations support these conclusions as well; the identity of the π-system substituent alters the δ–δ* (HOMO–LUMO) gap by up to 0.4 eV, whereas regioisomerism yields smaller changes in the electronic structure.

 Please wait while we load your content...
Please wait while we load your content...