Preparation and characterization of a novel Wells–Dawson heteropolyacid-based magnetic inorganic–organic nanohybrid catalyst H6P2W18O62/pyridino-Fe3O4 for the efficient synthesis of 1-amidoalkyl-2-naphthols under solvent-free conditions
Abstract
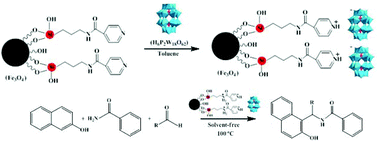
A novel magnetic inorganic–organic nanohybrid material H6P2W18O62/pyridino-Fe3O4 (HPA/TPI-Fe3O4) was fabricated and performed as an efficient, eco-friendly, and highly recyclable catalyst for the solvent-free, one-pot, and multi-component synthesis of various substituted 1-amidoalkyl-2-naphthols from the reaction of β-naphthol, an aldehyde, and benzamide with good to excellent yields (47–94%) and in a short span of time (25–60 min). The nanohybrid catalyst was prepared by the chemical anchoring of Wells–Dawson heteropolyacid H6P2W18O62 onto the surface of modified Fe3O4 nanoparticles with N-[3-(triethoxysilyl)propyl]isonicotinamide (TPI) linker. The magnetically recoverable catalyst was easily recycled at least eight times without any loss of catalytic activity. XRD, TEM, UV-vis, and FTIR confirmed that the heteropolyacid H6P2W18O62 is well dispersed on the surface of the solid support and its structure is retained after immobilization on the pyridine modified Fe3O4 nanoparticles. This protocol is developed as a safe and convenient alternate method for the synthesis of 1-amidoalkyl-2-naphthols utilizing an eco-friendly, and a highly reusable catalyst.

 Please wait while we load your content...
Please wait while we load your content...