Eco-friendly synthesis of bio-additive fuels from renewable glycerol using nanocrystalline SnO2-based solid acids†
Abstract
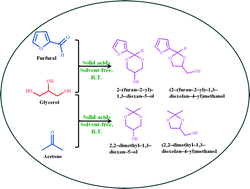
The present work has been undertaken with an aim to synthesize valuable bio-additive fuels from glycerol acetalization using SnO2-based solid acids. Various promoters, namely SO42−, MoO3 and WO3 were incorporated to the SnO2 using a wet-impregnation method. An extensive physicochemical characterization has been achieved by means of XRD, BET surface area, BJH analysis, FT-IR, pyridine adsorbed FT-IR, NH3-TPD, ICP-OES and XPS techniques. The BET surface area of SnO2 is significantly improved from 11 to 32, 56 and 41 m2 g−1 after the addition of the WO3, MoO3, and SO42− promoters, respectively. The XPS studies revealed that Sn is present in the +4 oxidation state, whereas Mo, W and S are in the +6 oxidation state in the prepared samples. In addition, the SO42−/SnO2 sample contained super acidic sites, along with strong- and medium-acidic sites. The amount of acidic sites was found to be 46.47, 61.81, 81.45 and 186.98 μmol g−1 for the SnO2, WO3/SnO2, MoO3/SnO2, and SO42−/SnO2 samples, respectively. The pyridine adsorbed FT-IR studies revealed the existence of a superior quantity of Brønsted acidic sites than Lewis acidic sites in the synthesized catalysts. Promoted SnO2 catalysts exhibited a promising catalytic performance for glycerol acetalization with acetone and furfural, and the activity of the catalysts was found to increase in the following order: SnO2 < WO3/SnO2 < MoO3/SnO2 < SO42−/SnO2. The outstanding performance of the SO42−/SnO2 catalyst is mainly due to the existence of a large amount of acidic sites associated with the super acidic sites. The achieved optimum glycerol conversions with acetone and furfural were ~98 and 99% over the SO42−/SnO2 catalyst, respectively.

 Please wait while we load your content...
Please wait while we load your content...