Insights into the active surface species formed on Ta2O5 nanotubes in the catalytic oxidation of CO
Abstract
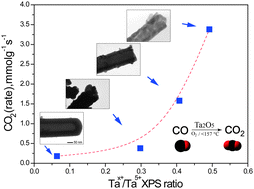
Freestanding Ta2O5 nanotubes were prepared by an anodizing method. As-anodized amorphous nanotubes were calcined at high temperature to obtain a crystalline phase. All materials were studied by means of BET analysis, XRD, TEM, SEM, XPS, and FTIR and were evaluated in the catalytic oxidation of CO. An XPS study confirmed the formation of different tantalum surface species after high temperature treatment of amorphous Ta2O5 nanotubes. Calcination at 800 °C generated Ta4+ while higher temperature (1000 °C) treatment led to the formation of Ta3+ species. These materials also showed significant differences in catalytic activity. Higher activity was observed for samples calcined at 800 °C than at 1000 °C, suggesting that Ta4+ species are active sites for CO oxidation.

 Please wait while we load your content...
Please wait while we load your content...