Conformer-selective photoelectron spectroscopy of α-lactalbumin derived multianions in the gas phase†
Abstract
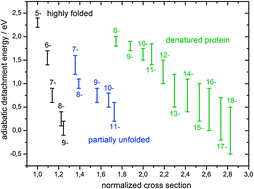
We have recorded conformer-selective, gas-phase photoelectron spectra of α-lactalbumin derived multianions generated by electrospraying solutions of both the native protein and its denatured form (as prepared by breaking the sulfur–sulfur bonds by chemical reduction). Three different groups of gas-phase multianion conformers have been observed and characterized. Highly-folded and partially-unfolded structures are obtained from solutions of the native protein. Only highly-elongated conformers are observed upon electrospraying the denatured protein. Adiabatic detachment energies were determined at several negative charge states for each conformer group. In comparison to highly-elongated conformations, highly-folded structures show a steeper decrease of electron binding energy with increasing negative charge. By comparing experimental detachment energies for highly-elongated structures with the predictions of a simple electrostatic model calculation, we have determined the effective dielectric shielding constant.

 Please wait while we load your content...
Please wait while we load your content...