Carbon-coated LiCrTiO4 electrode material promoting phase transition to reduce asymmetric polarization for lithium-ion batteries
Abstract
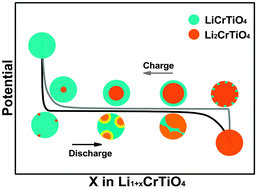
In this work, carbon-free and carbon-coated spinel LiCrTiO4 oxides were synthesized by a conventional solid state reaction. The lithium-ion diffusion coefficient and electronic conductivity of prepared electrode materials were systematically investigated using the galvanostatic intermittent titration technique (GITT), cyclic voltammetry (CV), and electrochemical impedance spectroscopy (EIS). The rate performances of the prepared materials were evaluated by galvanostatic charge–discharge. Carefully comparing the charge–discharge polarization potential of both materials, we unexpectedly discovered that the pristine LiCrTiO4 electrode demonstrated asymmetric polarization during the charging–discharging process, which is possibly attributed to the nonuniform electron conductivity between the endmember of a two-phase reaction, whereas carbon coating could level this phenomenon. Additionally, using an asymmetric core–shell model from the microscopic point of view can easily explain this common phenomenon. Meanwhile, this new research perspective can be extended to other active materials in lithium ion batteries.

 Please wait while we load your content...
Please wait while we load your content...