Selective staining of Brønsted acidity in zeolite ZSM-5-based catalyst extrudates using thiophene as a probe†
Abstract
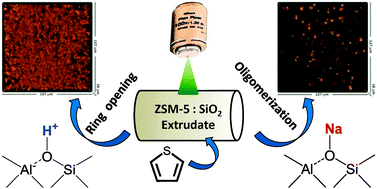
Optical absorption and confocal fluorescence micro-spectroscopy were applied to investigate Brønsted acidity in millimetre-sized extrudates of Na(H)-ZSM-5 and SiO2 with varying ZSM-5 content. Partially (residual Na present) and fully proton-exchanged extrudates were employed, using thiophene oligomerization as a probe reaction. Time-resolved in situ optical absorption spectra and time dependent DFT calculations revealed several initial reaction pathways during the oligomerization reaction. In particular, it was found that protonated thiophene monomers reacted by either oligomerization (via reaction with un-reacted thiophene monomers) or ring-opening, depending on the Brønsted acid site density in each sample. Moreover, fully-exchanged extrudates not only have significantly higher reactivity than partially-exchanged samples, but they also favour the formation of ring-opening products, that are not formed on the partially-exchanged samples. Confocal fluorescence microscopy was employed to visualise non-invasively in 3D, the heterogeneity and homogeneity of thiophene oligomers on partially- and fully-exchanged extrudates, respectively. Furthermore, it was observed that extrudates with high binder content produce a higher relative amount of conjugated species, related with a higher quantity of available monomer in the binder, which is able to react further with intermediates adsorbed on active sites. Moreover, these conjugated species appear to form near the external surface of ZSM-5 crystals/agglomerates.

 Please wait while we load your content...
Please wait while we load your content...