The origin of high electrolyte–electrode interfacial resistances in lithium cells containing garnet type solid electrolytes†
Abstract
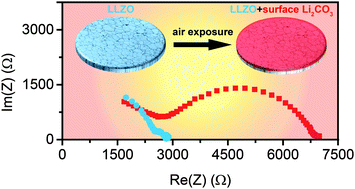
Dense LLZO (Al-substituted Li7La3Zr2O12) pellets were processed in controlled atmospheres to investigate the relationships between the surface chemistry and interfacial behavior in lithium cells. Laser induced breakdown spectroscopy (LIBS), scanning electron microscopy (SEM), X-ray diffraction (XRD), Raman spectroscopy, synchrotron X-ray photoelectron spectroscopy (XPS) and soft X-ray absorption spectroscopy (XAS) studies revealed that Li2CO3 was formed on the surface when LLZO pellets were exposed to air. The distribution and thickness of the Li2CO3 layer were estimated by a combination of bulk and surface sensitive techniques with various probing depths. First-principles thermodynamic calculations confirmed that LLZO has an energetic preference to form Li2CO3 in air. Exposure to air and the subsequent formation of Li2CO3 at the LLZO surface is the source of the high interfacial impedances observed in cells with lithium electrodes. Surface polishing can effectively remove Li2CO3 and dramatically improve the interfacial properties. Polished samples in lithium cells had an area specific resistance (ASR) of only 109 Ω cm2 for the LLZO/Li interface, the lowest reported value for Al-substituted LLZO. Galvanostatic cycling results obtained from lithium symmetrical cells also suggest that the quality of the LLZO/lithium interface has a significant impact on the device lifetime.


 Please wait while we load your content...
Please wait while we load your content...