Effect of enzymatic orientation through the use of syringaldazine molecules on multiple multi-copper oxidase enzymes
Abstract
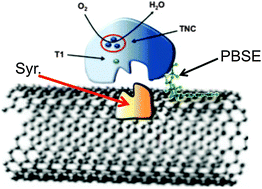
The effect of proper enzyme orientation at the electrode surface was explored for two multi-copper oxygen reducing enzymes: Bilirubin Oxidase (BOx) and Laccase (Lac). Simultaneous utilization of “tethering” agent (1-pyrenebutanoic acid, succinimidyl ester; PBSE), for stable enzyme immobilization, and syringaldazine (Syr), for enzyme orientation, of both Lac and BOx led to a notable enhancement of the electrode performance. For Lac cathodes tested in solution it was established that PBSE–Lac and PBSE–Syr–Lac modified cathodes demonstrated approximately 6 and 9 times increase in current density, respectively, compared to physically adsorbed and randomly oriented Lac cathodes. Further testing in solution utilizing BOx showed an even higher increase in achievable current densities, thus BOx was chosen for additional testing in air-breathing mode. In subsequent air-breathing experiments the incorporation of PBSE and Syr with BOx resulted in current densities of 0.65 ± 0.1 mA cm−2; 2.5 times higher when compared to an unmodified BOx cathode. A fully tethered/oriented BOx cathode was combined with a NAD-dependent Glucose Dehydrogenase anode for the fabrication of a complete enzymatic membraneless fuel cell. A maximum power of 1.03 ± 0.06 mW cm−2 was recorded for the complete fuel cell. The observed significant enhancement in the performance of “oriented” cathodes was a result of proper enzyme orientation, leading to facilitated enzyme/electrode interface interactions.

 Please wait while we load your content...
Please wait while we load your content...