Spectral evidence for hydrogen-induced reversible segregation of CO adsorbed on titania-supported rhodium†
Abstract
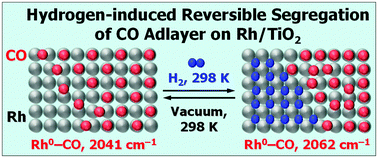
The reduction of a 1.3% Rh/TiO2 sample with carbon monoxide leads to the formation of uniform Rh nanoparticles with a mean diameter of dp ≈ 2.2 nm. Adsorption of CO on the reduced Rh/TiO2 produces linear and bridged carbonyls bound to metallic Rh0 sites and only a few geminal dicarbonyls of RhI. The ν(CO) of linear Rh0–CO complexes is strongly coverage dependent: it is observed at 2078 cm−1 at full coverage and at ca. 2025 cm−1 at approximated zero coverage. At low coverage, this shift is mainly caused by a dipole–dipole interaction between the adsorbed CO molecules while at high coverage, the chemical shift also becomes important. Hydrogen hardly affects the CO adlayer at high CO coverages. However, on a partially CO-covered surface (θCO ≈ 0.5), the adsorption of H2 at increasing pressure leads to a gradual shift in the band of linear Rh0–CO from 2041 to 2062 cm−1. Subsequent evacuation almost restores the original spectrum, demonstrating the reversibility of the hydrogen effect. Through the use of 12CO + 13CO isotopic mixtures, it is established that the addition of hydrogen to the CO–Rh/TiO2 system leads to an increase in the dynamic interaction between the adsorbed CO molecules. This evidences an increase in the density of the adsorbed CO molecules and indicates segregation of the CO and hydrogen adlayers. When CO is adsorbed on a hydrogen-precovered surface, the carbonyl band maximum is practically coverage independent and is observed at 2175–2173 cm−1. These results are explained by a model according to which CO successively occupies different rhodium nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...