Effect of pH on the chemical modification of quercetin and structurally related flavonoids characterized by optical (UV-visible and Raman) spectroscopy†
Abstract
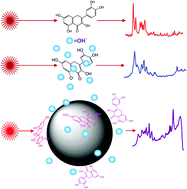
In this work we report the study of the chemical modifications undergone by flavonoids, especially by quercetin (QUC), under alkaline conditions by UV-visible absorption, Raman and surface-enhanced Raman scattering (SERS) spectroscopy, the study was performed in aqueous solution and also on Ag nanoparticles (AgNPs). Several processes are involved in the effect of alkaline pH both in solution and on AgNPs: autoxidation affecting mainly the C-ring of the molecule and giving rise to the molecular fragmentation leading to simpler molecular products, and/or the dimerization and further polymerization leading to species with a higher molecular weight. In addition, there exists a clear structure–instability correlation concerning mainly particular groups in the molecule: the C3–OH group in the C-ring, the catechol moiety in the B-ring and the C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C3 bond also existing in the C-ring. QUC possesses all these groups and exhibits high instability in alkaline solution. The SERS spectra registered at different pH revealed a change in the dimerization protocol of QUC going from the A- and C-rings-like-condensation to B-ring-like-condensation. Increasing the knowledge of the chemical properties of these compounds and determining the structure–activity relationship under specific environmental factors allow us to improve their beneficial properties for health as well as the preservation of Cultural Heritage objects, for example, by preventing their degradation.
C3 bond also existing in the C-ring. QUC possesses all these groups and exhibits high instability in alkaline solution. The SERS spectra registered at different pH revealed a change in the dimerization protocol of QUC going from the A- and C-rings-like-condensation to B-ring-like-condensation. Increasing the knowledge of the chemical properties of these compounds and determining the structure–activity relationship under specific environmental factors allow us to improve their beneficial properties for health as well as the preservation of Cultural Heritage objects, for example, by preventing their degradation.

 Please wait while we load your content...
Please wait while we load your content...