Highly fluorinated naphthalenes and bifurcated C–H⋯F–C hydrogen bonding†
Abstract
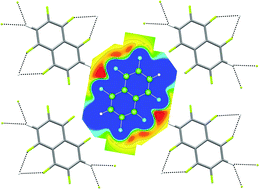
The synthesis and crystal structures of 1,2,4,5,6,8-hexafluoronaphthalene and 1,2,4,6,8-pentafluoronaphthalene are reported. Intermolecular interactions are dominated by offset stacking and by C–H⋯F–C hydrogen bonds. For hexafluoronaphthalene, molecules are linked in layers with (4,4) network topology via R12(6) C–H⋯(F–C)2 supramolecular synthons that are rationalised by consideration of the calculated electrostatic potential of the molecule. Such an arrangement is prevented by the additional hydrogen atom in pentafluoronaphthalene and molecules instead form tapes via an R12(8) (C–H⋯F)2 synthon. The geometric characteristics of C–H⋯(F–C)2 bifurcated hydrogen bonds have been analysed for crystal structures in the Cambridge Structural Database (6416 crystal structures; 9534 C–H⋯(F–C)2 bifurcated hydrogen bonds). A geometric analysis of these hydrogen bonds has enabled the extent of asymmetry of these hydrogen bonds to be assessed and indicates a preference for symmetrically bifurcated interactions.


 Please wait while we load your content...
Please wait while we load your content...