The phosphorescent co-crystals of 1,4-diiodotetrafluorobenzene and bent 3-ring-N-heterocyclic hydrocarbons by C–I⋯N and C–I⋯π halogen bonds†
Abstract
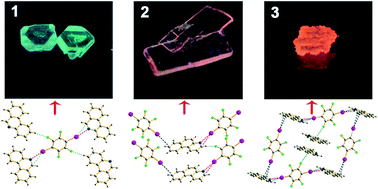
The co-crystals 1–3 were successfully assembled from 1,4-diiodotetrafluorobenzene (1,4-DITFB) and bent 3-ring-N-heterocyclic hydrocarbons (3-R-NHHs) based on halogen bonds (XBs) and other weak interactions. The halogen bond as a good strategy of introducing a metal-free heavy-atom perturber at a suitable stoichiometry is further confirmed, which makes the spin–orbital coupling more efficient and the phosphorescence more observable. Herein, the selected 3-R-NHHs in co-crystals 1–3 show different emissions, of green, orange-yellow and orange phosphorescence with well defined vibrational bands at 544 nm (max) for 1, 592 nm (max) for 2 and 605 nm (max) for 3, whereas the pure 3-R-NHHs are not phosphorescent in the solid state. The reason can be mainly ascribed to the different N-positions within the three 3-R-NHHs that affect the C–I⋯π halogen bond properties, which influence further the energy level of the excited triplet state and meanwhile make the transition from T1 to the higher vibrational level of the S0 state become more probable with respect to the free monomer. It is estimated that in the co-crystals the energy level of the excited ππ* triplet state is lowered, as evidenced by the presence of a bathochromic effect in the maximum emission bands of 50–100 nm. The other interactions and local molecular environment can affect the phosphorescent behaviours, too. The different phosphorescent behaviours of three co-crystals should be suitable for the design of some appropriate π-type acceptors for the development of phosphorescent materials via the introduction of halogen bonds.

 Please wait while we load your content...
Please wait while we load your content...