Water channels and zipper structures in Schiff base-like Cu(ii) and Ni(ii) mononuclear complexes†
Abstract
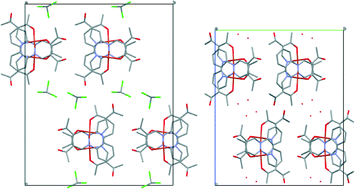
The crystal structures of four Cu(II) complexes and one Ni(II) complex bearing a square planar N2O2 coordination sphere are discussed. In all cases a distorted square planar coordination sphere is observed that is independent of the metal centre and the ligand and is only influenced by the packing of the molecules in the crystal. For the less distorted copper complexes metal–aromatic interactions are observed, while no significant intermolecular interactions are found for the nickel complexes. This can be explained by the different electronic character of Cu(II) and Ni(II) and is also reflected in the differences in the solvatochromism of these complexes.

 Please wait while we load your content...
Please wait while we load your content...