Osteogenic differentiation of human mesenchymal stem cells on α5 integrin binding peptide hydrogels is dependent on substrate elasticity†
Abstract
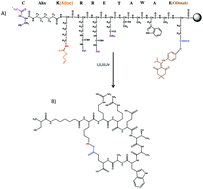
The extracellular matrix plays a crucial role in controlling human mesenchymal stem cell (hMSC) biology including differentiation, and α5β1 integrin signaling plays an important role during osteogenic differentiation of hMSCs. Here, peptide-functionalized hydrogels were used to examine the role of α5β1 integrin signaling in inducing osteogenic differentiation in hMSCs. Further, the role of substrate elasticity was also studied. A thiol–ene chemistry was used to functionalize poly(ethylene glycol) hydrogels with a pendant peptide moiety, c(RRETAWA), as previous studies have shown that RRETAWA containing peptides bind to the α5β1 integrin with very high specificity. Notably, hMSC attachment to c(RRETAWA)-functionalized hydrogels was found to occur primarily through α5 integrins, as the number of attached cells was significantly reduced to ∼20% upon blocking the α5 integrin during culture. To investigate the interplay between stiffness and c(RRETAWA) concentration, hydrogels were formulated with Young's moduli of ∼2 kPa (soft) and ∼25 kPa (stiff) and c(RRETAWA) concentrations of 0.1 mM and 1 mM. Stiff substrates led to ∼3.5 fold higher hMSC attachment and ∼3 fold higher cell area in comparison to soft substrates. hMSCs formed robust and larger focal adhesions on stiff substrates at 1 mM c(RRETAWA) compared to soft substrates. Alkaline phosphatase (ALP) activity in hMSCs cultured on stiff gels at 0.1 mM and 1 mM c(RRETAWA) was increased 2.5 and 3.5 fold, respectively after 14 days in growth media. hMSCs did not show an increase in ALP activity when cultured on soft gels. Further, gene expression of osteogenic related genes, core binding factor-1, osteopontin and Collagen-1a at day 14 in hMSCs cultured on stiff gels at 1 mM c(RRETAWA) were increased 10, 7 and 4 fold, respectively, while on soft gels, gene expression was at basal levels. Collectively, these results demonstrate that the combination of high substrate stiffness and α5β1 integrin signaling stimulated by c(RRETAWA) is sufficient to induce osteogenic differentiation of hMSCs without requiring the addition of soluble factors.

 Please wait while we load your content...
Please wait while we load your content...