Synthesis, cytotoxicity and bioimaging of a novel Hg2+ selective fluorogenic chemosensor†
Abstract
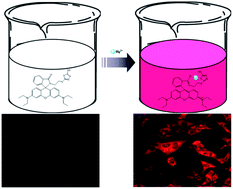
A rhodamine B derivative 2 with a terminal mercapto thiadiazole moiety was successfully synthesized and applied for selective recognition of Hg2+ in aqueous–acetonitrile (1 : 1, v/v, pH 7) solution by employing its photophysical properties. Low toxicity and precise cell-permeability of the ligand were used to probe in vitro intracellular mercury contamination using L-929 cells (mouse fibroblast cells) and BHK-21 (hamster kidney fibroblast) through a confocal fluorescence microscopic experiment and bio-imaging results showed the equal applicability of the ligand toward both tested cell lines. Meanwhile, it exhibited distinct Hg2+ induced increment in the fluorescence and absorption intensities with induction of an apparent colorimetric change from colorless to reddish pink, providing naked eye mercury detection based on the metal-promoted intramolecular electronic rearrangement in the probe molecule. The probe responds selectively to Hg2+ over various competitive cations (Sc3+, Yb3+, In3+, Ce3+, Sm3+, Cr3+, Sn2+, Pb2+, Fe2+, Ni2+, Co2+, Cu2+, Ba2+, Ca2+, Mg2+, Ag+, Cs+, Cu+, and K+) with marked fluorogenic response and selective colorimetric changes with high sensitivity of 30 nM. We expect that the proposed method will serve as a practical tool for environmental sample analysis and biological studies.

 Please wait while we load your content...
Please wait while we load your content...