Colorimetric copper(ii) ion sensor based on the conformational change of peptide immobilized onto the surface of gold nanoparticles†
Abstract
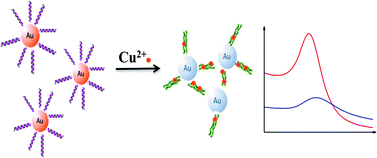
Detection of heavy metal ions has attracted great attention, and the colorimetric assay has remarkable advantages, such as convenient, efficient, free-equipment and always visible. Herein, we report a highly sensitive and selective colorimetric sensor for the determination of copper(II) ions based on the conformational change of Cu2+-binding peptides immobilized onto the surface of gold nanoparticles. In the presence of copper(II) ions, the peptides modified gold nanoparticles (p-AuNPs) will cooperatively bind together, resulting in aggregation and precipitation of the p-AuNPs, thus color change can be observed from wine red to colorless. Nevertheless, other cations like Mg2+, Ca2+, Zn2+, Fe3+, Pb2+, Mn2+, Ba2+, Ni2+, Co2+, K+ and Ag+ cannot have such an effect, so no obvious disturbance occurs in the same concentration to Cu2+. With this well-designed sensing platform, the detection range of copper(II) ions is found to be 10–150 μM, which falls into the maximum accepted level of 1.3 ppm (∼20 μM) as set by the US Environmental Protection Agency. Moreover, modification of AuNPs with differential binding peptides may provide new and simple platforms for the detection of other heavy metal ions, so the strategy proposed for the detection of Cu2+ in this work can be extended for more applications in the future.

 Please wait while we load your content...
Please wait while we load your content...