Multimodal optical characterisation of collagen photodegradation by femtosecond infrared laser ablation†
Abstract
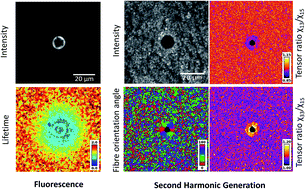
Collagen is a structural component of the human body, as a connective tissue it can become altered as a result of pathophysiological conditions. Although the collagen degradation mechanism is not fully understood, it plays an important role in ageing, disease progression and applications in therapeutic laser treatments. To fully understand the mechanism of collagen alteration, in our study photo-disruptive effects were induced in collagen I matrix by point-irradiation with a femtosecond Ti-sapphire laser under controlled laser ablation settings. This was followed by multi-modal imaging of the irradiated and surrounding areas to analyse the degradation mechanism. Our multi-modal methodology was based on second harmonic generation (SHG), scanning electron microscope (SEM), autofluorescence (AF) average intensities and the average fluorescence lifetime. This allowed us to quantitatively characterise the degraded area into four distinct zones: (1) depolymerised zone in the laser focal spot as indicated by the loss of SHG signal, (2) enhanced crosslinking zone in the inner boundary of the laser induced cavity as represented by the high fluorescence ring, (3) reduced crosslinking zone formed the outer boundary of the cavity as marked by the increased SHG signal and (4) native collagen. These identified distinct zones were in good agreement with the expected photochemical changes shown using Raman spectroscopy. In addition, imaging using polarisation-resolved SHG (p-SHG) revealed both a high degree of fibre re-orientation and a SHG change in tensor ratios around the irradiation spot. Our multi-modal optical imaging approach can provide a new methodology for defining distinct zones that can be used in a clinical setting to determine suitable thresholds for applying safe laser treatments without affecting the surrounding tissues. Furthermore this technique can be extended to address challenges observed in collagen based tissue engineering and used as a minimally invasive diagnostic tool to characterise diseased and non-diseased collagen rich tissues.

 Please wait while we load your content...
Please wait while we load your content...