Mass spectrometric investigation of the role of the linking polypeptide chain in DNA polymerase I
Abstract
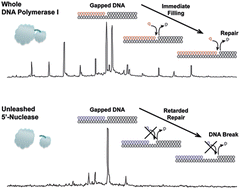
DNA polymerase I offers great promise for a wide range of biotechnological applications due to its capability to add labeled nucleotides into double-stranded large DNA molecules by using both polymerase and nuclease domains. Accordingly, it is crucially important to thoroughly characterize this enzyme for further developments. Although the enzyme has been thus far characterized using mainly traditional analytical instruments, here we utilized an advanced and convenient means of mass spectrometry to elucidate enzymatic functions and mechanisms by measuring DNA oligomers generated by polymerase and nuclease reactions. Our analysis revealed several novel enzymatic features, including the observation that polymerase readily dissociates from the DNA molecules containing a wide single-stranded section. From this finding, we reasoned a serious situation of DNA break because polymerase domains cannot efficiently repair the wide single-stranded section, which is susceptible to DNA breaks. Furthermore, we deduced a plausible explanation for a paradoxical question as to why two domains of polymerase and 5′-nuclease are linked by a small and flexible polypeptide in polymerase I. The polypeptide link seems to prevent a 5′-nuclease from causing DNA breaks by locating a polymerase domain closely for immediate repair reaction. Here we present experimental evidence to prove our hypothesis via a set of mass spectrometric analyses as well as single DNA molecule observation and bacterial cell growth assay. Consequently, mass spectrometric analysis for DNA polymerase I provides a meaningful biological insight that a polypeptide link can be a molecular leash to control an aggressive domain in order to prevent unmanageable damages.

 Please wait while we load your content...
Please wait while we load your content...