Submicron silica spheres decorated with silver nanoparticles as a new effective sorbent for inorganic mercury in surface waters†
Abstract
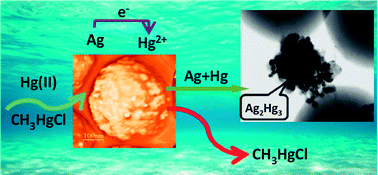
An analytical method using silica supported silver nanoparticles as a novel sorbent for the enrichment and determination of inorganic mercury (iHg) in surface water samples has been developed. Silver nanoparticles (AgNPs) were synthesized by a completely green procedure and were deposited onto the amine functionalized surface of silica submicrospheres (SiO2–NH2). The prepared nanocomposite material (SiO2/AgNPs) was characterized by transmission electron microscopy, UV-vis spectroscopy, X-ray diffraction and atomic force microscopy. The sorption and desorption characteristics of the nanosorbent SiO2/AgNPs toward Hg species were investigated by a batch method. An excellent separation of iHg and methylHg was achieved in 20 minutes at pH 2. The high selectivity of the SiO2/AgNPs toward iHg was explained by Hg(II) reduction and subsequent silver–mercury amalgam formation. The analytical procedure for the enrichment and determination of inorganic mercury in surface waters was developed based on solid phase extraction and ICP-MS measurements. The total Hg content was determined after water sample mineralization. The recoveries reached for iHg in different surface waters e.g. river and Black sea water samples varied from 96–101%. The limits of quantification are 0.002 μg L−1 and 0.004 μg L−1 for iHg and total Hg, respectively; the relative standard deviations varied in the ranges of 5–9% and 6–11% for iHg and total Hg, respectively, for Hg content from 0.005 to 0.2 μg L−1. The accuracy of the procedure developed for total Hg determination was confirmed by a comparative analysis of surface river (ICP-MS) and sea (CV AFS) waters.

 Please wait while we load your content...
Please wait while we load your content...