Tunable filling rate and increased ferromagnetic properties of nickel-filled carbon nanotubes synthesized from a Pauli paramagnetic lanthanum nickel (LaNi5) alloy catalyst†
Abstract
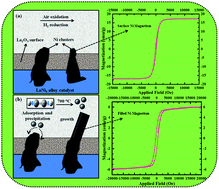
Ferromagnetic nickel-filled carbon nanotubes (Ni-filled CNTs) were synthesized by chemical vapor deposition using Pauli paramagnetic lanthanum nickel alloy (LaNi5) both as a catalyst and as a source for the Ni-filling. The effect of various catalyst oxidation temperatures on the tunable filling rate and coercivity of Ni-filled CNTs were investigated. The influence of the addition of catalyst oxidation step prior to growth is required for filling of metallic Ni in CNTs. The filling rate and the magnetic properties of Ni-filled CNTs were determined by transmission electron microscopy (TEM) and vibrating sample magnetometer, respectively. TEM results showed that the filling rate of CNTs strongly depended on the catalyst oxidation temperatures and the higher Ni-filling was found to be at 500 °C. With the increase of catalyst oxidation temperatures, partially filled CNTs structure (at 300 °C) changed to Ni nanowire encapsulated CNTs (at 400 and 500 °C), then to fine Ni nanoparticles situated on the surface of CNTs (at 600 °C). The Ni-filling process through the catalyst oxidation is explained using the “surface segregation” mechanism. Magnetic hysteresis loops of Ni-filled CNTs revealed that their coercivity depended on the content and size of the encapsulated Ni. Coercivity value of Ni-filled CNTs synthesized after the catalyst oxidation temperature at 400 °C reached the maximum at 446 Oe, which is significantly larger than that of the bulk Ni at room temperature. Thus our work demonstrates a simple and facile approach toward tunable filling rate and ferromagnetic properties of Ni-filled CNTs would have potential in various applications.

 Please wait while we load your content...
Please wait while we load your content...