Metal ion-assisted reshaping of Cu2O nanocrystals for catalytic applications†
Abstract
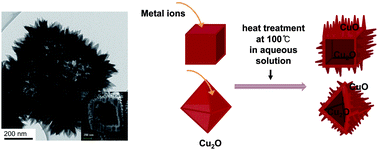
Copper oxides have received much attention because of their potential in energy-related applications such as gas-phase reactions, photoreactions, and batteries. In this study, the surfaces of cube- and octahedron-shaped Cu2O nanocrystals are transformed into thorny CuO, which have larger surface areas and better stability in an oxidizing environment. When the shaped Cu2O crystals were treated in an aqueous solution at 100 °C with metal ions such as Pd, Pt, and Au, the crystals became hollow particles, retaining their original shape with CuO thorns on the surface. Addition of the metal ions initiated the transformation by galvanic replacement, and then the protons generated in the replacement catalyzed sequent dissolution of Cu2O and deposition of CuO. The resulting particles showed enhanced activity and stability for CO oxidation.

 Please wait while we load your content...
Please wait while we load your content...