Realization of both high hydrogen selectivity and capacity in a guest responsive metal–organic framework†
Abstract
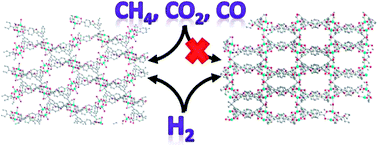
Two newly designed semi-flexible tetratopic carboxylate ligands, 5′,5′′′′-(propane-2,2-diyl)bis(2′-methoxy-[1,1′:3′,1′′-terphenyl]-4,4′′-dicarboxylate) (pbtd-OMe4−) and 5′,5′′′′-(propane-2,2-diyl)bis(2′-ethoxy-[1,1′:3′,1′′-terphenyl]-4,4′′-dicarboxylate) (pbtd-OEt4−), have been used to connect dicopper paddlewheel building units to afford two isostructural metal–organic frameworks, Cu2(H2O)2(pbtd-OR)·xS (R = Me, PCN-38·xS; R = Et, PCN-39·xS, S represents noncoordinated guest molecules, PCN = porous coordination network) with novel structure and gas sorption properties upon activation. PCN-39 undergoes structural transformations upon guest solvent removal, leading to observation of distinct phases from in situ powder X-ray diffraction measurements, and exhibits selective adsorption of H2 (up to 2.0 wt%) over CO, CO2, and N2, which can be explained by optimized space-filling of the pendant ethoxy group. PCN-38 undergoes no transformation upon activation and exhibits hydrogen uptake up to 2.2 wt%, as well as moderate uptake of other gases. The selective adsorption of hydrogen over other gases highlights the potential application of PCN-39 in industrially important gas separation.

 Please wait while we load your content...
Please wait while we load your content...