Carbon nanotube channels selectively filled with monodispersed Fe3−xO4 nanoparticles
Abstract
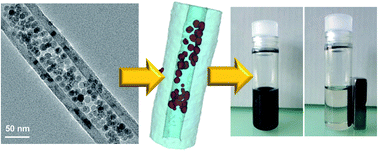
Magnetic iron oxide (Fe3−xO4) nanoparticles (NPs) with high density and narrow size distribution were selectively cast inside or at the outer surface of the channels of carbon nanotubes (CNTs) by performing thermal decomposition of an organic iron precursor in liquid-phase medium in the presence of adequately pre-treated CNTs. The size of the Fe3−xO4 NPs generated by the synthesis is around 13 ± 3 nm and no agglomeration was observed thanks to the presence of oleic acid playing the role of surfactant in the synthesis medium. The selective filling of the Fe3−xO4 NPs inside the CNT channel was confirmed by X-ray diffraction, transmission electron microscopy (also in tomography mode) and X-ray photoelectron spectroscopy. Such NPs were found to be less sensitive towards oxidation compared to the same NPs synthesized without CNTs according to the powder X-ray diffraction and they display thus a higher saturation magnetization (65 emu g−1). Such a composite was found to be superparamagnetic at room temperature. Due to its new “magnetic state”, it could efficiently be employed in applications, which need both magnetic and electric properties, especially in catalysis in liquid-phase medium where the catalyst–product separation is facilitated by the magnetic properties of the catalyst.

 Please wait while we load your content...
Please wait while we load your content...