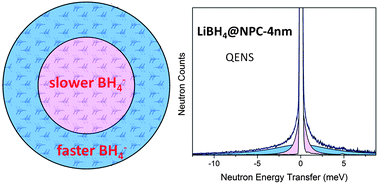
Particle size and particle–framework interactions have profound effects on the kinetics, reaction pathways, and even thermodynamics of complex hydrides incorporated in frameworks possessing nanoscale features. Tuning these properties may hold the key to the utilization of complex hydrides in practical applications for hydrogen storage. Using carefully synthesized, highly-ordered, nanoporous carbons (NPCs), we have previously shown quantitative differences in the kinetics and reaction pathways of LiBH4 when incorporated into the frameworks. In this paper, we probe the anion mobility of LiBH4 confined in NPC frameworks by a combination of solid state NMR and quasielastic neutron scattering (QENS) and present some new insights into the nanoconfinement effect. NMR and QENS spectra of LiBH4 confined in a 4 nm pore NPC suggest that the BH4− anions nearer the LiBH4–carbon pore interface exhibit much more rapid translational and reorientational motions compared to those in the LiBH4 interior. Moreover, an overly broadened BH4− torsional vibration band reveals a disorder-induced array of BH4− rotational potentials. XRD results are consistent with a lack of LiBH4 long-range order in the pores. Consistent with differential scanning calorimetry measurements, neither NMR nor QENS detects a clear solid–solid phase transition as observed in the bulk, indicating that borohydride–framework interactions and/or nanosize effects have large roles in confined LiBH4.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...