Role of photoexcited electrons in hydrogen evolution from platinum co-catalysts loaded on anatase TiO2: a first-principles study
Abstract
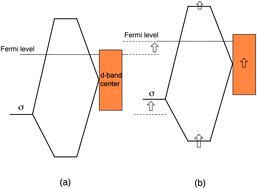
The mechanism of photocatalytic reactions that occur at platinum co-catalysts loaded on TiO2 is not well understood at the atomic scale. The photoexcited electrons that are generated in TiO2 should play an important role in the evolution of hydrogen by water splitting on a platinum surface. Our density-functional calculations reveal that electrons introduced in the conduction band of TiO2 contribute to an upward shift of the Fermi level of platinum via accumulation at the Pt/TiO2 interface, which results in weakened adsorption of H2 molecules on the Pt surface. We study how the electronic structure of the Pt/TiO2 interface is modified by photoexcited electrons which are represented by extra electrons introduced into the conduction band by substituting fluorine for oxygen. One important consequence of doping is that bonding–antibonding splitting associated with the hybridization of H2-sigma and Pt-d states is narrowed, which weakens the H2–Pt binding and enhances the evolution of H2.

 Please wait while we load your content...
Please wait while we load your content...