Template-free synthesis of hollow α-Fe2O3 microcubes for advanced lithium-ion batteries
Abstract
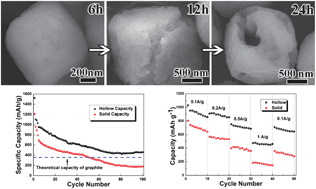
Hollow α-Fe2O3 microcubes were fabricated by a facile hydrothermal method in an ethanol–water co-solvent system. The as-synthesized microcubes have a uniform size with an edge length of about 1.5 μm. Time and solvent proportion dependent experiments reveal that the ethanol adsorption and surface-protected etching mechanisms play key roles in the formation hollow cubic structures. Compared with their solid counterparts, hollow α-Fe2O3 microcubes show an enhanced electrochemical performance in terms of long-term cycling (458 mA h g−1 at a current density of 100 mA g−1 after 100 cycles) and high rate capability (859, 855, 688 and 460 mA h g−1 at current densities of 100, 200, 500 and 1000 mA g−1, respectively). These remarkable electrochemical properties can be attributed to the unique hollow microstructure, which could retain structural stability, relieve stress and increase reaction areas.

 Please wait while we load your content...
Please wait while we load your content...