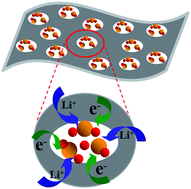
The commercialization of lithium–sulfur (Li–S) batteries has so far been limited by their rapid capacity fading, which is induced by dissolution of intermediate polysulfides and the pulverization of the sulfur cathode due to volume expansion. Herein, we reported an efficient strategy to confine active sulfur in chemically tailored graphene nanosheets, which were prepared via modified chemical activation of hydrothermal reduced graphene oxide hydrogels. Due to its high specific surface area, large pore volume, controllable size and distribution of nanopores, the two-dimensional (2D) highly porous activated graphene nanosheets (AGNs) were proved to be a promising scaffold to uniformly confine elemental sulfur (S) in their nanopores with high loading. The resultant AGNs/S nanocomposites exhibited a reversible capacity up to 1379 mA h g−1 at 0.2 C as well as remarkable cycling stability, which may contribute to the desirable structural features. The dense nanopores of AGNs, as “micro-reactors” for the electrochemical reactions of sulfur, minimized polysulfide dissolution and shuttling in the electrolyte, and also reserved fast transport of lithium ions to the sequestered sulfur by ensuring good electrolyte penetration. Furthermore, the AGNs with good electronic conductivity allowed good transport of electrons from/to the poorly conducting sulfur for electrochemical reactions at high rates.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...