Independent control of water retention and acid–base pairing through double-shelled microcapsules to confer membranes with enhanced proton conduction under low humidity
Abstract
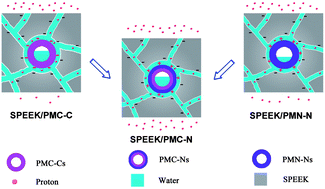
Proton exchange membranes (PEM) with affordable and controllable proton conductivity under low humidity are crucial to the commercial application of PEM fuel cells. In this study, double-shelled polymer microcapsules bearing a carboxylic acid inner shell and imidazole outer shell (PMC-Ns) are synthesized via distillation–precipitation polymerization and then incorporated into a sulfonated poly(ether ether ketone) matrix to fabricate composite membranes. The inner shell renders the absorbed water of lower chemical potential and higher bound water content, yielding the composite membrane with enhanced water retention properties. The higher water uptake ensures composite membrane facilitated proton transfer via a vehicle mechanism and the lower water loss confers a reduced proton conductivity decline. The outer shell generates sulfonic acid–imidazole pairs within the membranes, which construct low-energy-barrier pathways to facilitate proton transfer via the Grotthuss mechanism. Under identical conditions, PMC-Ns endow much higher proton conductivity to composite membranes than the microcapsules with both carboxylic acid inner shell and outer shell, or both imidazole inner shell and outer shell. Particularly, incorporating 20 wt% PMC-Ns affords the composite membrane a 1.7 times increase in proton conductivity under 100% relative humidity (RH) and a 41.8 times increase in proton conductivity under 20% RH. Moreover, the methanol barrier property of the composite membranes is explored.

 Please wait while we load your content...
Please wait while we load your content...