Mechanistic control over morphology: self-assembly of a discotic amphiphile in water
Abstract
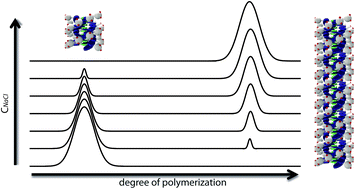
We report on the self-assembly of discotic amphiphiles that contain chelated gadolinium(III) ions and are based on the C3-symmetrical benzene-1,3,5-tricarboxamide motif. Fluorescence spectroscopy, SAXS and cryo-TEM experiments demonstrate that a bimodal distribution of small and large aggregates is formed in a ratio that is dependent on the ionic strength. The results correlate with the previously reported degree of cooperativity of the polymerization mechanism, which increases with increasing NaCl concentration. Hence, by tuning the electrostatic interactions between the ligands at the periphery we can tune the cooperativity of the self-assembly. This tunability provides a versatile handle to adjust the size and shape of the discotic amphiphiles, which have potential as supramolecular MRI contrast agents.

 Please wait while we load your content...
Please wait while we load your content...