Reversible encapsulation of lysozyme within mPEG-b-PMAA: experimental observation and molecular dynamics simulation†
Abstract
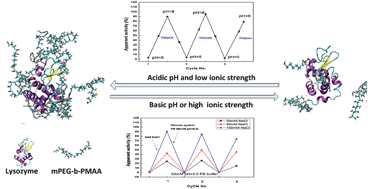
We synthesized a novel hydrophilic, negatively charged block polymer composed of polyethylene glycol (PEG) and poly(methacrylic acid) (PMAA) using atom-transfer radical polymerization (ATRP). The encapsulation of a positively charged protein, represented by hen-egg white lysozyme, by mPEG-b-PMAA micelles was achieved using a pH or salt-concentration swing, as shown by both structural characterization using dynamic light scattering and transmission electron microscopy and an activity assay. All-atom molecular dynamics simulations showed that using an acidic pH gave a more compact polymer–micelle assembly than did using a basic pH. As a result, this compact structure had less solvent-accessible surface area (SASA), indicating that lysozyme was encapsulated by mPEG-b-PMAA and that the active site was shielded by the polymer. This made the active site less accessible to the substrate. These accounted for the low apparent activity at an acidic pH in our experiments. A neutral or basic pH intensified the electrostatic repulsive interaction, which prevented the formation of polymer–lysozyme complex. The molecular simulation indicated that encapsulation of lysozyme by the polymer micelles could be divided into two consecutive steps. The first step involved the attachment of the negatively charged polymer chain to the positively charged portion of lysozyme, driven by electrostatic attractive force. Then, the hydrophobic interaction between the polymer and lysozyme became dominant and led to a more compact assembly with a reduced energy state. These simulations agreed with our experimental observations and provided molecular insight helpful for the design, fabrication, and application of protein-incorporated polymer micelles.

 Please wait while we load your content...
Please wait while we load your content...