Well-defined biodegradable amphiphilic conetworks†
Abstract
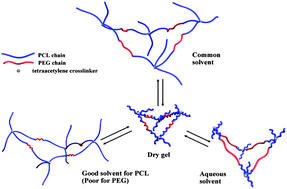
A series of amphiphilic conetworks (APCNs) with well-defined molecular structures were prepared via a copper-catalyzed 1,3-dipolar azide–alkyne cycloaddition (CuAAC) of tetrakis(2-propynyloxymethyl)methane (TMOP), diazide end-functionalized triblock copolymers of poly(ε-caprolactone) with poly(ethylene glycol) (N3-PCL-PEG-PCL-N3). The so-prepared APCNs exhibit unique properties of ordered nanophase separation of hydrophilic (HI) and hydrophobic (HO) phases, and a variable swelling capacity both in water and organic solvent. The morphology, surface properties and thermal behavior of the APCNs were investigated by scanning electron microscopy (SEM), water contact angle (WCA), and differential scanning calorimetry (DSC), respectively. The physical properties of APCNs depended on the ratio of HI–HO, which can be regulated via precise synthesis of N3-PCL-PEG-PCL-N3. The analysis of an in vitro cell viability assay suggests that the APCNs have excellent biocompatibility. The prepared APCNs are excellent carriers for controlled drug release. The hydrophilic choline theophyllinate and hydrophobic 5-fluorouracil (5-FU) were loaded into the APCNs simultaneously as model drugs to study the release from APCNs. The well-controlled drug release is attributable to the well-defined molecular structure and tunable HI/HO composition of the APCNs.

 Please wait while we load your content...
Please wait while we load your content...