Monitoring degradation of matrix metalloproteinases-cleavable PEG hydrogels via multiple particle tracking microrheology
Abstract
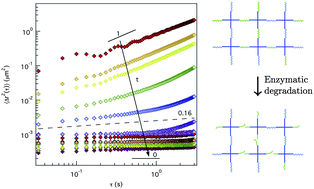
The design of hydrogel matrices for cell encapsulation and tissue regeneration has become increasingly complex. Oftentimes, researchers seek to recapitulate specific biophysical and biochemical cues critical for the resident cell population and an in depth understanding of changes in the local microstructure and rheological properties of the synthetic matrix during enzymatic degradation would be extremely beneficial. Multiple particle tracking microrheology (MPT) enables simultaneous characterization of rheological properties and visualization of the microstructure in an evolving hydrogel scaffold. MPT measures the Brownian motion of fluorescently labeled probe particles embedded in the material, which is directly related to rheological properties using the Generalized Stokes–Einstein Relation (GSER). Here, we study a hydrogel scaffold consisting of a four-arm poly(ethylene glycol) (PEG) end functionalized with norbornene that is cross-linked with both a nondegradable PEG–dithiol and a matrix metalloproteinase (MMP) degradable peptide sequence (KCGPQG↓IWGQCK) using thiol–ene chemistry. The material degradation is measured as a function of time and extent of degradability, focusing on measuring the gel–sol transition. Using time–cure superposition, we determine the critical degradation time and critical extent of degradability for this specific gel formulation as tc = 1.85 h and pc = 0.589, respectively, and the critical relaxation exponent, n = 0.16. Finally, spatial information gained by MPT measurements quantifies the heterogeneity within the scaffold showing that these hydrogels degrade homogeneously when collagenase is introduced in solution at a concentration of 0.1–0.3 mg mL−1. Understanding the microstructural and rheological properties of a material near the gel–sol transition enables researchers to improve their insight as to how cells remodel their microenvironment when encapsulated in gels, and more precisely design and manipulate this parameter to improve three-dimensional culture systems.

 Please wait while we load your content...
Please wait while we load your content...