Synthesis of a well-defined glycoconjugate vaccine by a tyrosine-selective conjugation strategy†
Abstract
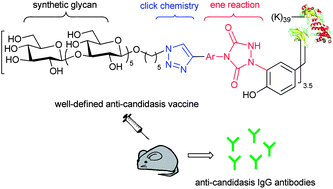
An anti-candidiasis glycoconjugate vaccine was prepared via a tyrosine-selective alkynylation and a click chemistry mediated glycoconjugation sequence. It features a well-defined glycan,

 Please wait while we load your content...
Please wait while we load your content...