Remarkable coordination behavior of alkyl isocyanides toward unsaturated vicinal frustrated P/B Lewis pairs†
Abstract
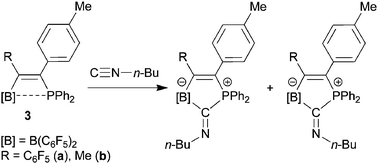
The conjugated frustrated phosphane/borane Lewis pairs formed by 1,1-carboboration of a substituted diphenylphosphino acetylene, undergo a synergistic 1,1-addition reaction to n-butyl isocyanide with formation of new B–C and P–C bonds to the former isonitrile carbon atom. Using tert-butyl isocyanide dynamic behaviour between the isocyanide–[B] adduct and the 1,1-addition product formation was observed in solution. The different modes of isocyanide binding to the FLPs in the solid state were characterized using X-ray crystal structure analyses and comprehensive 11B and 31P solid-state magic-angle-spinning (MAS-) NMR experiments. The free FLP, the Lewis adduct at the borane group, and the cyclic product resulting from isocyanide addition to both reaction centers, can be differentiated via11B and 31P isotropic chemical shifts, 11B nuclear electric quadrupole coupling constants, isotropic indirect 11B–31P spin–spin coupling constants, and 11B⋯31P internuclear distances measured by rotational echo double resonance.

 Please wait while we load your content...
Please wait while we load your content...