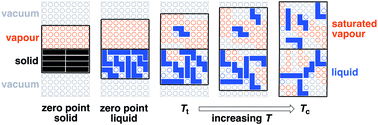
The bulk properties of liquids provide information on the thermodynamic properties of intermolecular interactions between non-polar molecules. Literature data on noble gases, alkanes and perfluorocarbons have been analysed to investigate the relationship of the magnitude of van der Waals interactions between non-polar molecules with chemical structure and molecular architecture. A molecular model of the liquid state is proposed based on the concept of a zero point void, which has a volume of 5 Å3 and a surface area of 19 Å2 per molecule. This void is the minimum volume required for each molecule to independently exchange an intermolecular contact and hence move in the liquid. Three hypothetical constructs (the zero point solid, the zero point liquid and the ideal liquid) are used to separate the processes of melting, thermal expansion and evaporation. Melting is a complex process affected by contributions from changes in structure in the solid, formation of the zero point void and thermal expansion. Evaporation involves breaking all intermolecular interactions between a molecule and the surrounding liquid and therefore provides a straightforward measure of the total van der Waals interaction. Experimental data for the enthalpy barrier to the evaporation process indicate that the total van der Waals interaction is approximately a linear function of molecular surface area, worth 0.3 kJ mol−1 Å−2 for non-polar liquids. Expansion involves breaking local intermolecular contacts and is generally associated with a substantially lower energy barrier that depends on the degree of coupling between interaction sites across the molecular surface. The expansion process is closely related to the phase changes that occur at the triple point and the critical point, and these properties are therefore a complex function of molecular architecture. Similarly, the free energy change for the vapour–liquid equilibrium depends on the molecular architecture. In contrast, transfer free energy data for liquid–liquid equilibria suggest that van der Waals interactions are a simple function of molecular surface area, independent of atom type or molecular architecture. For processes that take place within the liquid state, free energy changes due to exchange of van der Waals interactions are therefore expected to be small.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...