4,5-Dihaloimidazolium-based ionic liquids: effects of halogen-bonding on crystal structures and ionic conductivity†
Abstract
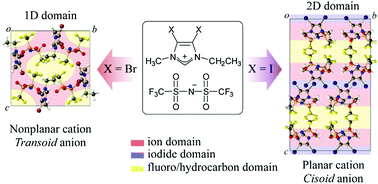
Halogen substitutions at the 4- and 5-positions of the imidazolium cation cause characteristic molecular arrangements in the crystal phase and increase the number of ion pairs in the liquid phase because of halogen-bonding between the halide groups and the counter anions.

 Please wait while we load your content...
Please wait while we load your content...