A g-C3N4/BiOBr visible-light-driven composite: synthesis via a reactable ionic liquid and improved photocatalytic activity†
Abstract
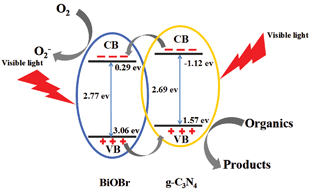
g-C3N4/BiOBr composite photocatalysts have been synthesized in the presence of the reactable ionic liquid 1-hexadecyl-3-methylimidazolium bromide ([C16mim]Br). The as-prepared samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FT-IR), diffuse reflectance spectroscopy (DRS), photoluminescence (PL) spectroscopy, electrochemical impedance spectroscopy (EIS), and photocurrent analysis. During the reaction process, the ionic liquid [C16mim]Br acted as solvent, reactant, template and dispersing agent at the same time, leading the g-C3N4 to disperse well on the surface of the BiOBr flower-like microspheres. The photocatalytic ability of the as-prepared photocatalysts was evaluated using rhodamine B (RhB) as a target pollutant. The photocatalysts exhibited a significantly enhanced photocatalytic performance in the degradation of RhB. The results of PL, EIS, and photocurrent tests indicated that g-C3N4 combined and dispersed well on the surface of BiOBr which facilitated electron–hole separation, and led to the increased photocatalytic activity. The optimal g-C3N4 content for the photocatalytic activity of the g-C3N4/BiOBr composites was determined. Radical trap experiments certified that the hole was the main reactive species for the photocatalytic degradation of RhB. A possible mechanism of g-C3N4 for the enhancement of visible light performance was proposed.

 Please wait while we load your content...
Please wait while we load your content...