Experimental and theoretical studies of tetramethoxy-p-benzoquinone: infrared spectra, structural and lithium insertion properties†
Abstract
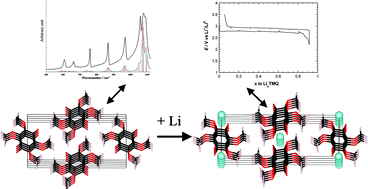
In the search for low-polluting
* Corresponding authors
a
LRCS-CNRS UMR 7314, Université de Picardie, 33, Rue Saint-Leu, 80039 Amiens, France
E-mail:
christine.frayret@u-picardie.fr
b IMN-CNRS UMR 6502, Université de Nantes, 2, Rue de la Houssinière, 44322 Nantes, France
c ISM-CNRS UMR 5255, 351 Cours de la Libération, 33405 Talence, France
d Department of Chemistry, Syracuse University, Syracuse, NY 13244, United States
e UCCS, équipe de Chimie du Solide - CNRS UMR 8181, ENSC Lille-UST Lille, Cité Scientifique - Bât. C7 - BP 80108, 59655 Villeneuve d'Ascq cedex, France
f CNR-ISTM, Istituto di Scienze e Tecnologie Molecolari, via Golgi 19, Milano, Italy
g School of Chemistry, Monash University, Clayton, Victoria 3800, Australia
h Réseau sur le Stockage Electrochimique de L’Energie (RS2E), FR CNRS 3459, France
In the search for low-polluting

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
G. Bonnard, A. Barrès, Y. Danten, D. G. Allis, O. Mentré, D. Tomerini, C. Gatti, E. I. Izgorodina, P. Poizot and C. Frayret, RSC Adv., 2013, 3, 19081 DOI: 10.1039/C3RA41974F
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
